Researchers reveal the assembling mechanism of the cytosolic AhR complex
Dioxin is an environmental pollutant with a strong biotoxicity, which is extremely harmful to human. When exposed to dioxin, human may suffer from the tumorigenesis and the destruction of immunity. The aryl hydrocarbon receptor (AhR) is the only one of the biomolecules in organisms that can recognize and bind dioxin. When dioxin is present at the cytoplasm, AhR can bind dioxin with a high affinity, which triggers the activation of the related signaling pathway to produce biological effects. Studies of the structure and function of AhR can help to understand the specific molecular mechanism of AhR signaling pathway, develop AhR-targeted drugs and develop high-sensitivity biosensors to detect dioxin-like environmental pollutants.
On January 16, 2023, a study from Wen et al. led by Prof. Fei Sun at the Institute of Biophysics, Chinese Academy of Sciences (CAS), entitled "Cryo-EM structure of the cytosolic AhR complex" was published on Structure. In this study, researchers purified mouse AhR cytosolic comlex and resolved the high resolution structure of the complex using cryo-EM. A novel interaction pattern in the complex and the mechanisms of ligand binding of AhR were revealed.
AhR is a ligand-activated transcription factor that belongs to the bHLH-PAS family, which harbours a bHLH domain, PAS-A domain and PAS-B domain. The PAS-B domain of AhR is responsible for reaction to different stimuli with a ligand binding pocket in the center. AhR can be activated by a variety of signals and is widely involved in various physiological processes such as cytotoxic regulation, cell proliferation and differentiation, neurodevelopment and immune response. In the classical AhR signaling pathway, AhR firstly forms a stable resting complex in the cytosol with chaperone Hsp90, cochaperone p23, and AhR interacting protein XAP2. The ligand-binding event will activate the related signaling pathway and regulate the expression of downstream genes. Although the signaling pathway is now roughly understood, the interaction pattern and assembling mechanism of the cytosolic AhR complex still remain unknown.
The study from Wen et al. successfully purified stable and homogeneous mouse AhR cytosolic complex by the screening and Optimization of the purification conditions. The structure of the cytosolic AhR complex, which consists of AhR, Hsp90, p23, XAP2, was resolved at a high resolution using cryo-EM, where the structure of the ligand-free state of AhR PAS-B domain in mammal was firstly resolved. The AhR PAS-B domain consists of four a-helices and five ?strands surrounding a ligand-binding pocket in the center. In this complex, AhR binds to Hsp90 not only by the residues in the PAS-B domain to form hydrophilic interactions, but also though a loop between PAS-A and PAS-B domains (which is named the bridge motif) to interact with the Hsp90 dimeric lumen. Besides Hsp90, AhR interacts with XAP2 through the C-terminal loop and has potential contacts with p23. AhR is fixed by these interactions and so that can AhR exist stably in the cytosol.
By structural comparisons between the resolved mouse AhR PAS-B domain and other PAS-B domains, this study proposed a potential mechanism of ligand-entry of mouse AhR. In the ligand-free state, the DE-loop of AhR presents a specific conformation, leaving a distinct entry in the PAS-B domain, which is available for ligand-entry. After ligand binding, the PAS-B ligand-binding pocket is expanded and the DE-loop undergoes conformational changes to block the entry. So that the ligand is captured inside the PAS-B pocket. AhR may specifically recognize and bind pollutant ligands through this proposed potential mechanism.
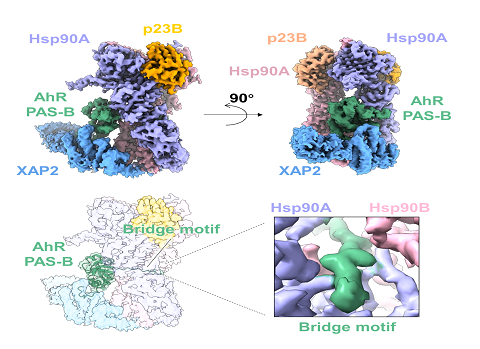
Figure. The schematic diagram of the cytosolic AhR complex structure, where AhR is colored green.
Prof. Yun Zhu, AProf. Yujia Zhai and Prof. Fei Sun from Institute of Biophysics, Chinese Academy of Sciences are the co-corresponding authors of this work. Dr. Zuoling Wen is the first author of this work. This work was supported by grants from the Ministry of Science and Technology of China and the Strategic Priority Research Program of the Chinese Academy of Sciences.
Link: https://pubmed.ncbi.nlm.nih.gov/36649707/
附件下载: